Light vs. Cancer: No Drugs, No Chemo — Inside the “Molecular Jackhammer” Breakthrough
Cancer treatment has always carried a heavy emotional weight. For many people, the word cancer immediately brings images of chemotherapy chairs, radiation rooms, surgical scars, painful side effects, hair loss, fear, and uncertainty. Even when modern cancer care saves lives, the journey can be physically and emotionally brutal. That is why every new cancer breakthrough captures public imagination so quickly—especially when it sounds almost impossible.
No drugs.
No chemotherapy.
No radiation in the traditional sense.
Just light.
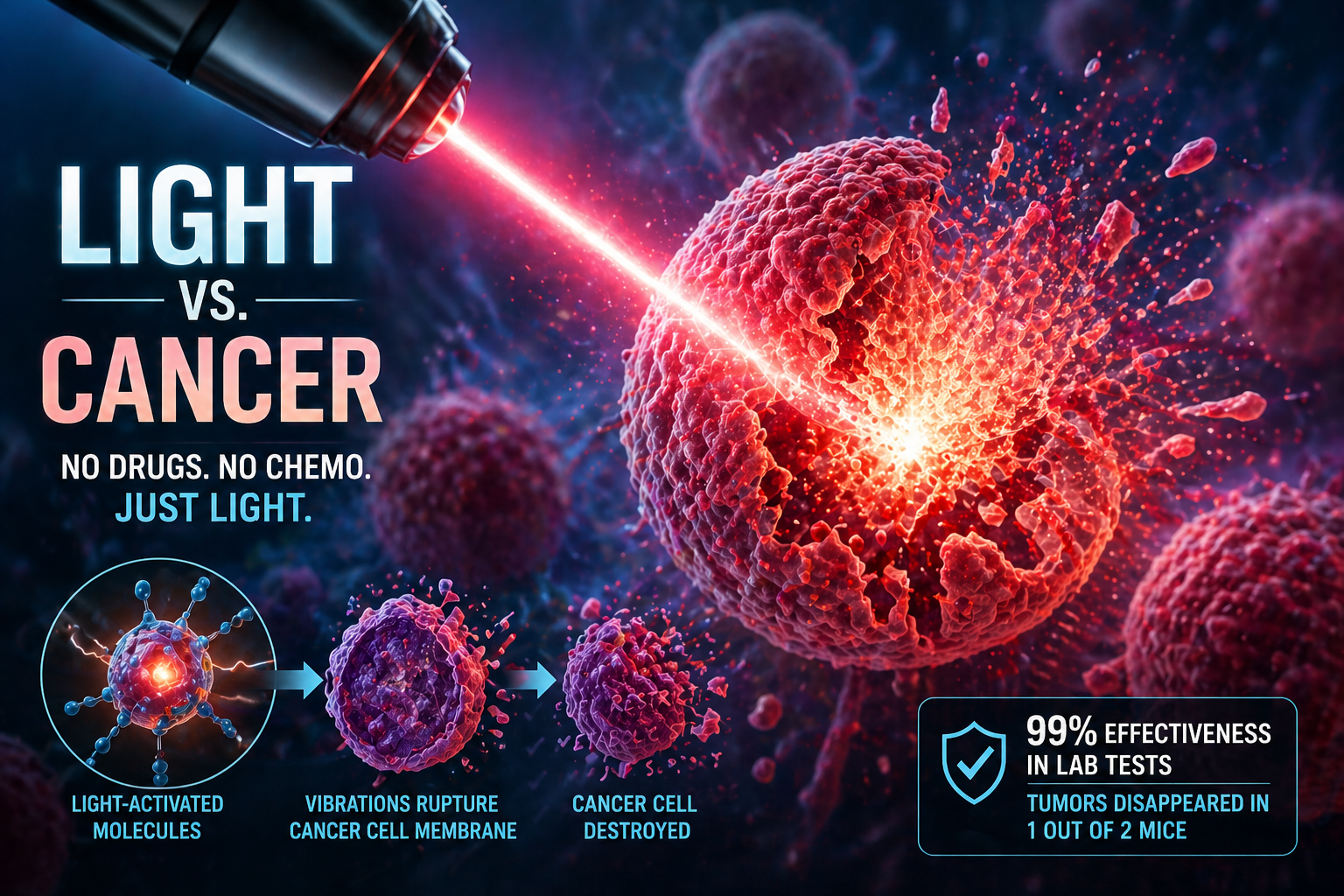
A team of researchers from Rice University and collaborating institutions reported a striking experimental method that uses near-infrared light to activate special molecules attached to cancer cell membranes. Once activated, these molecules vibrate with extreme force, physically disrupting and rupturing the cancer cell’s outer membrane. The researchers called these molecules “molecular jackhammers” because their movement acts less like a chemical poison and more like a nanoscale mechanical attack.
In lab experiments, the method destroyed 99% of human melanoma cells in vitro. In mouse studies, half of the treated animals became cancer-free after treatment. The study, published in Nature Chemistry, has been described by Rice University as a new way to eradicate cancer cells through light-induced whole-molecule vibration.
It is one of those scientific stories that feels like it belongs in the future: a cancer cell destroyed not by flooding the body with toxic chemicals, but by shaking its membrane apart from the outside.
But as exciting as this sounds, it must be understood carefully. This is not an approved cancer cure. It is not available as a clinical treatment. It has not yet been proven in humans. The results are early, experimental, and limited to lab-grown cells and animal models. Still, the idea is powerful because it opens a bold question: what if cancer could be attacked mechanically, precisely, and without relying on conventional drugs?
The Core Idea: Killing Cancer by Breaking Its Membrane
Every cell has a membrane. Think of it as a living border wall: flexible, selective, and essential. It controls what enters and leaves the cell. It helps the cell communicate with its surroundings. It protects the internal machinery that keeps the cell alive.
If that membrane is badly damaged, the cell cannot survive.
Traditional cancer therapies often work through chemical or biological pathways. Chemotherapy drugs may interfere with DNA replication or cell division. Immunotherapy tries to help the immune system recognize and attack tumors. Targeted therapies block specific molecular signals that cancer cells rely on. Radiation damages DNA and other cellular structures.
The molecular jackhammer method is different.
Instead of poisoning the cell or blocking a signaling pathway, researchers used small dye molecules called aminocyanines, which are already known in medical imaging contexts. These molecules can attach to cell membranes. When exposed to near-infrared light, their atoms vibrate in a synchronized way. The vibration is so intense that it can physically tear open the membrane of a cancer cell. Rice University described this as the activation of a plasmon-like vibration in the whole molecule, leading to membrane rupture.
This is why the “jackhammer” analogy works so well. A construction jackhammer does not melt concrete or poison it. It breaks it through repeated mechanical force. At a vastly smaller scale, these molecules appear to create a similar effect against cancer cell membranes.
Why Near-Infrared Light Matters
The use of near-infrared light is one of the most important parts of the story.
Visible light does not travel very deeply into human tissue. That is why shining a regular flashlight on your skin only illuminates the surface. Near-infrared light, however, can penetrate deeper into tissue than visible light. It is already used in several imaging and biomedical applications because it interacts with the body in useful ways.
In this experimental therapy, near-infrared light acts as the trigger. The molecules are not simply floating around and destroying cells randomly. They need the light stimulus to activate their intense vibration. That gives researchers a potential level of control: deliver or localize the molecules, shine near-infrared light on the target area, and activate the mechanical effect where it is needed.
This is one reason scientists are so interested in the technique. It suggests the possibility of treating certain tumors with precision, using light as the switch.
Of course, this also raises practical challenges. Not every tumor is easy to reach with light. Skin cancers and surface-accessible tumors may be more straightforward than deep tumors inside the pancreas, brain, lungs, or abdomen. For deeper cancers, researchers would need safe and effective ways to deliver light internally, possibly through fiber optics, endoscopy, interventional procedures, or other medical technologies.
So while near-infrared light gives the method an advantage, it does not magically solve every delivery problem.
What the Study Actually Showed
The headline number—99% effectiveness—is dramatic, but context matters.
The researchers tested the molecular jackhammer method on human melanoma cells grown in lab cultures. In that controlled in vitro environment, the method killed 99% of the cancer cells. They also tested the approach in mice with melanoma tumors. In those animal experiments, 50% of treated mice became cancer-free. Texas A&M, one of the collaborating institutions, summarized the work in similar terms, noting the 99% killing efficiency in vitro and cancer-free outcomes in half of the treated mice.
These results are impressive. But there is a huge difference between killing cancer cells in a dish and curing cancer in a human body.
Lab-grown cells are simplified systems. They do not fully reproduce the complexity of a human tumor. Real tumors are surrounded by blood vessels, immune cells, connective tissue, pressure gradients, oxygen differences, and protective microenvironments. Cancer cells also vary from patient to patient and even within the same tumor. A therapy that works beautifully in a dish may become far less effective in a living human body.
Mouse studies are more informative than cell cultures, but they are still not human trials. Many cancer therapies that look promising in mice never become successful treatments for people. Sometimes they fail because of toxicity. Sometimes because the treatment cannot reach tumors effectively. Sometimes because the biology does not translate. Sometimes because the human immune system, tumor environment, or disease stage changes everything.
That is why the most accurate interpretation is this: the molecular jackhammer method is a fascinating preclinical breakthrough, not a proven cancer therapy.
Why This Method Is So Different From Chemotherapy
Chemotherapy is often described as a “systemic” treatment because it travels throughout the body. This can be lifesaving when cancer has spread, but it also explains many of its side effects. Chemotherapy targets fast-dividing cells, and while cancer cells divide rapidly, so do some healthy cells—such as those in hair follicles, the digestive tract, and bone marrow.
The molecular jackhammer concept is different because it is not mainly based on chemical toxicity. It is based on mechanical disruption. In theory, that could allow a more localized attack if the molecules and light can be directed toward cancer cells.
This is why the discovery feels so revolutionary. A cancer cell membrane is not being attacked because a drug blocks a biochemical pathway. It is being attacked because the molecule attached to it is physically vibrating with destructive force.
The researchers also distinguished this from photothermal therapy and photodynamic therapy. Photothermal therapy uses light to create heat that kills cells. Photodynamic therapy uses light-activated chemicals to generate reactive oxygen species that damage cells. The molecular jackhammer method works through a different mechanism: synchronized molecular vibration strong enough to rupture membranes.
That distinction matters. Cancer cells can sometimes evolve resistance to chemical drugs by changing transport pumps, repairing DNA damage, altering receptors, or bypassing blocked pathways. Mechanical membrane rupture may be harder for a cell to “outsmart” in the same way—although that still needs much more study.
The Beauty of a Mechanical Attack
One of the most intriguing aspects of this research is philosophical as much as scientific. It reframes cancer treatment as a physical engineering problem.
What if cancer cells can be destroyed not only through chemistry, genetics, or immunity, but through motion?
At the nanoscale, molecules are not still. They rotate, vibrate, bend, twist, and respond to energy. Modern chemistry increasingly explores how molecular movement can be controlled and exploited. Molecular machines have already won Nobel-level attention. The idea of using tiny molecular structures to perform mechanical work inside biological systems is one of the most exciting frontiers in medicine.
The molecular jackhammer approach belongs to that frontier.
It imagines a therapeutic future where molecules can be placed at a disease site and activated like microscopic tools. Instead of merely delivering a drug, the molecule itself becomes a machine. It moves. It shakes. It breaks.
That is why this discovery feels larger than one cancer study. It points toward a future of mechanically activated medicine.
Why Melanoma Was the Test Case
The study focused on melanoma, a dangerous form of skin cancer. Melanoma is a logical early target for light-based therapies because skin tumors are more accessible to light than many internal cancers. This does not mean the method will only apply to melanoma, but it does mean melanoma is a practical place to begin.
Melanoma is also clinically important. While many cases can be treated successfully when caught early, advanced melanoma can be aggressive and life-threatening. Modern immunotherapies and targeted therapies have transformed melanoma treatment in recent years, but not all patients respond, and resistance can develop.
A light-activated mechanical approach could eventually become another weapon in the melanoma treatment arsenal—especially if it can be combined safely with existing therapies.
But again, that is a future possibility, not a current clinical option.
The “No Drugs” Claim Needs Careful Wording
The phrase “no drugs, no chemo” is emotionally powerful, but scientifically it needs nuance.
This technique does not use conventional chemotherapy drugs. It does not work like standard drug-based cancer therapy. However, it does involve introducing special molecules into the biological system. These aminocyanine molecules are not “drugs” in the traditional chemotherapy sense, but they are still therapeutic agents if used for treatment. They must be delivered, distributed, activated, metabolized, and cleared safely.
So the better phrase may be:
No traditional chemotherapy. No chemical poisoning mechanism. A light-activated molecular therapy using mechanical force.
That may be less viral, but it is more accurate.
Precision matters because cancer patients are often vulnerable to exaggerated claims. A headline that says “light cures cancer without drugs” can easily become misleading. The truth is more interesting and more responsible: researchers are exploring a new class of light-activated molecular machines that may mechanically destroy cancer cells.
That is still extraordinary.
What Must Happen Before Human Use?
Before this technology can become a real treatment, several major questions must be answered.
First, researchers must show that the method is safe. Destroying cancer cell membranes is useful only if healthy cells are spared or protected. Scientists need to understand how the molecules distribute in the body, whether they accumulate in healthy tissues, how long they remain active, and whether they trigger inflammation, immune reactions, toxicity, or unexpected damage.
Second, researchers must solve delivery. Getting the molecules to the right tumor cells is one challenge. Getting near-infrared light to the right tissue depth is another. A therapy for a skin tumor may require a different delivery system than a therapy for a liver tumor or brain tumor.
Third, the technique must be tested across cancer types. Melanoma is only one cancer. Breast cancer, colon cancer, pancreatic cancer, lung cancer, prostate cancer, ovarian cancer, leukemia, lymphoma, and brain cancers all behave differently. A method that works in one cancer model may not work equally well in others.
Fourth, researchers must determine whether tumors return. Full remission in some mice is encouraging, but long-term recurrence, metastasis, and survival need deeper investigation.
Finally, the treatment must go through human clinical trials. These trials typically begin with safety-focused Phase 1 studies, then move toward dose optimization, efficacy testing, comparison with existing standards, and regulatory review.
That process can take years.
Why the Breakthrough Still Matters
Even with all these limitations, the discovery matters because it expands the imagination of cancer therapy.
Cancer treatment has already entered an era of precision. We now have targeted therapies, checkpoint inhibitors, CAR-T cell therapies, antibody-drug conjugates, radioligand therapies, and personalized genomic profiling. The molecular jackhammer concept fits into this broader movement toward more intelligent, more selective cancer treatment.
It also challenges the old assumption that cancer must always be attacked by biochemical toxicity. If mechanical molecular motion can kill cancer cells, then researchers may develop many other forms of mechanically triggered therapy.
Imagine future oncology tools that combine:
light activation,
tumor-targeting molecules,
imaging guidance,
immune stimulation,
localized treatment delivery,
and real-time response monitoring.
That is the kind of future this research hints at.
The Risk of Overhype
Every cancer breakthrough walks a dangerous line between hope and hype.
Hope is necessary. Patients and families need it. Scientists need it too. Hope drives funding, creativity, and persistence.
But hype can be harmful. It can make early research sound like an available cure. It can encourage desperate patients to delay proven treatment. It can feed conspiracy thinking, especially when people misunderstand why preclinical discoveries take so long to reach hospitals.
The molecular jackhammer study is not hype by itself. It is real research from credible institutions, published in a serious journal. The hype begins when people remove the context and present it as if cancer has already been defeated.
Cancer has not been defeated by light. Not yet.
What scientists have done is demonstrate a powerful new mechanism that may one day contribute to cancer therapy.
That difference matters.
Could This Replace Chemo?
In the near future, no.
If the technology succeeds, it is more likely to first become an additional tool for specific cancers or specific clinical situations. It might be used for accessible tumors, localized disease, or in combination with surgery, immunotherapy, or other treatments. It may help treat tumors that can be illuminated safely. It may become useful where conventional therapies fail or where lower-toxicity options are needed.
Replacing chemotherapy entirely is a much bigger claim.
Chemotherapy remains important because many cancers are systemic. Leukemia, lymphoma, metastatic disease, and microscopic cancer spread cannot always be treated by targeting one visible tumor. A light-activated local therapy would need sophisticated delivery strategies to address widespread disease.
So the future may not be “light instead of all cancer treatment.”
It may be “light as one part of smarter cancer treatment.”
A New Era of Cancer Therapy?
The most exciting thing about molecular jackhammers is not just the current result. It is the principle.
For decades, cancer medicine has asked:
Can we poison cancer more than we poison the patient?
Can we block the mutation?
Can we wake up the immune system?
Can we cut it out?
Can we burn it with radiation?
Now another question enters the room:
Can we shake cancer apart?
That question sounds almost poetic, but it is rooted in real chemistry and physics. It turns light into a trigger, molecules into machines, and cancer cell membranes into vulnerable structures.
If future studies confirm safety and effectiveness, this technique could become part of a new generation of non-traditional cancer therapies. It may help reduce dependence on harsh systemic treatments for certain cases. It may create new options for patients who cannot tolerate conventional therapy. It may also inspire other molecular-machine approaches far beyond cancer.
But for now, the responsible verdict is clear.
Final Verdict: A Stunning Breakthrough, Not a Cure Yet
The Rice University molecular jackhammer research is one of the most fascinating cancer science stories of recent years. It shows that near-infrared light can activate special molecules attached to cancer cell membranes, causing intense vibration that ruptures and kills the cells. In lab tests, the technique destroyed 99% of human melanoma cells. In mouse experiments, half of treated animals became cancer-free.
That is remarkable.
But it is not yet a human treatment. It is not proof that light can cure cancer in patients. It is not a reason to avoid chemotherapy, surgery, radiation, immunotherapy, or other evidence-based care. It is a promising preclinical discovery that needs careful testing, safety evaluation, and clinical trials.
Still, the idea is beautiful in its simplicity.
A molecule waits on the surface of a cancer cell.
A beam of near-infrared light arrives.
The molecule begins to vibrate.
The membrane fails.
The cancer cell dies.
No dramatic chemical storm. No classic chemotherapy mechanism. Just physics, chemistry, and light working together at the smallest scale.
Cancer medicine may not be entering a world without drugs or chemo tomorrow. But it may be entering a world where light becomes sharper, smarter, and more powerful than we ever imagined.